Coverage

Traditional Medicare Part B
No step edit or PA required
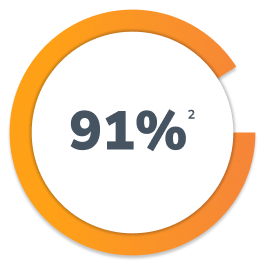
Commercial
No or only 1 step edit required
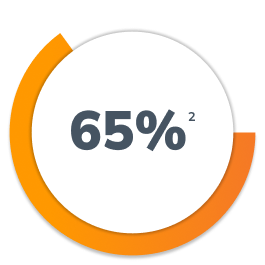
Medicare Advantage
No or only 1 step edit required
*Based on MMIT data 2022–2025 (and additional data sources since launch) based on medical benefit coverage. Data is verified internally.1
†Percentages shown utilize August 2025 MMIT coverage data for EVENITY® and IQVIA LAAD 2024 claims data. Data is verified against internally reported data for EVENITY® via the Organized Customer Team. Specifically, we verify formulary statuses for all known formulary decisions with MMIT, as well as covering institutions and downstream formularies periodically. This is intended to attenuate the risk of faulty data in this dataset.
‡All Medical Lives associated with Coverage Restriction for EVENITY® in MMIT coverage data snapshot as of August 2025 are included in this analysis.2
PA, prior authorization.













