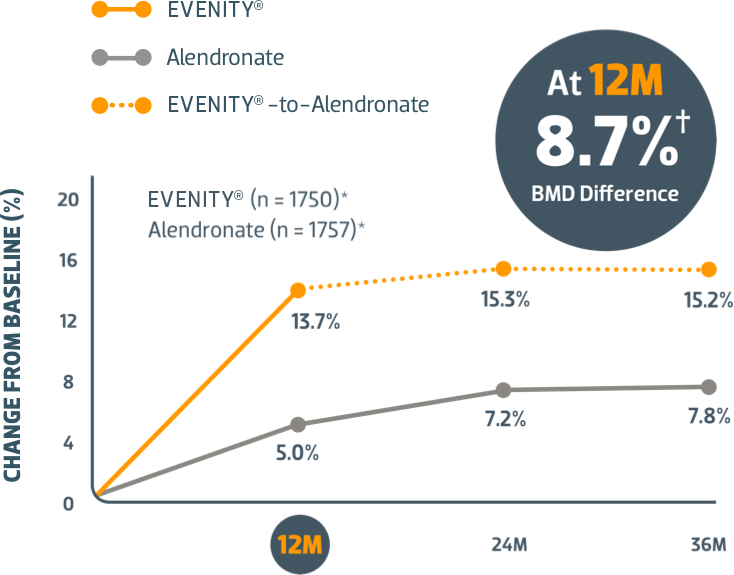
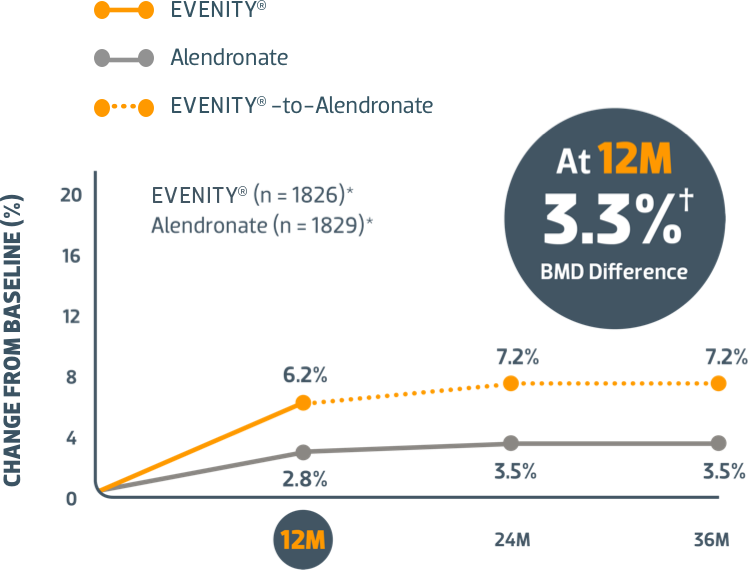
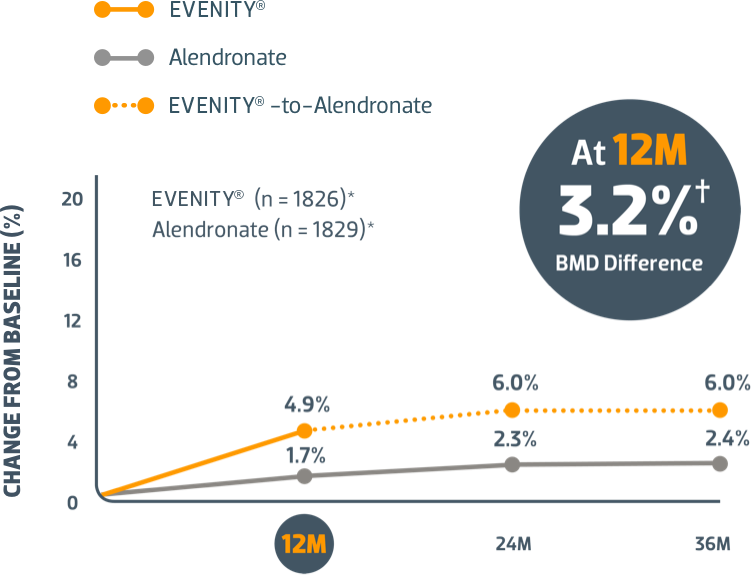
Start with EVENITY® first after fracture when your patients' risk of another is at its highest1,2
EVENITY® First Followed by Alendronate vs Alendronate Alone1,3
Fracture Risk Reduction
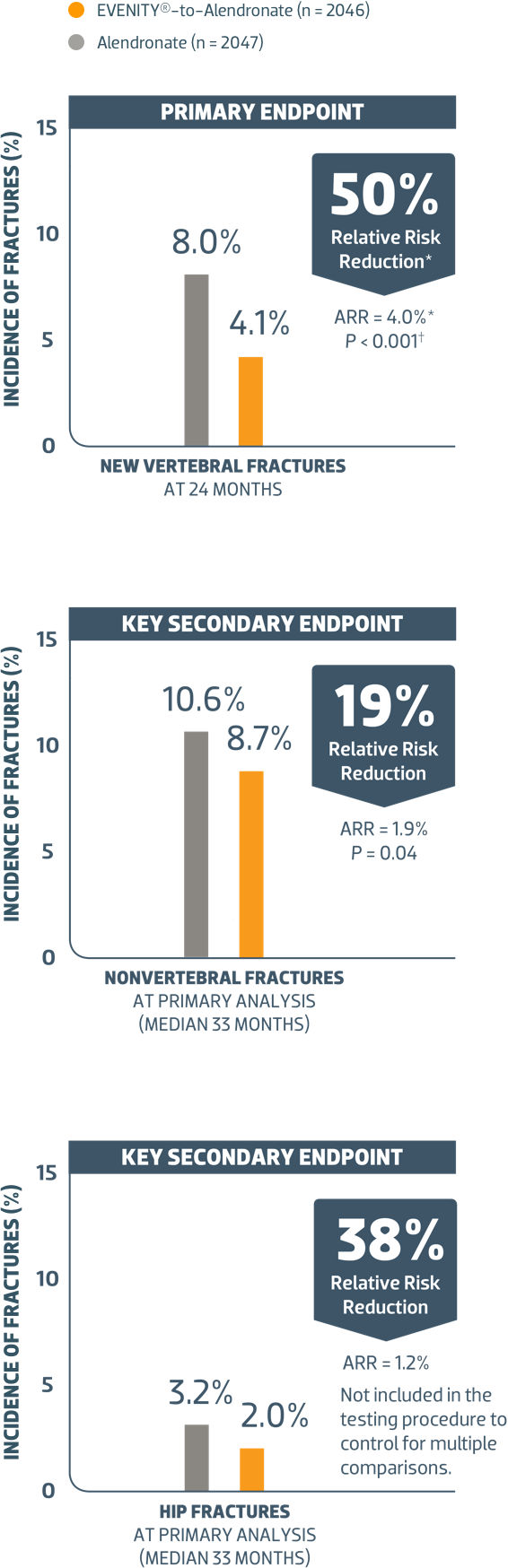
This was an event-driven trial and the duration of follow-up varied across subjects. The median duration of subject follow-up for the primary analysis period was 33 months1
EVENITY® for 12 months followed by alendronate reduced the incidence of clinical fracture (nonvertebral and symptomatic vertebral fracture) at primary analysis (median 33 months) (P < 0.001)1
*Absolute and relative risk reductions are based on the Mantel‑Haenszel method adjusting for age strata, baseline total hip BMD T‑score (≤ -2.5,>-2.5), and presence of severe vertebral fracture at baseline.
† P value based on logistic regression model (new vertebral fracture) or Cox proportional hazards model (other fracture types) adjusting for age strata, baseline total hip BMD T‑score, and presence of severe vertebral fracture at baseline.
‡Nonvertebral fractures excluded fractures of the skull, facial bones, metacarpals, fingers, and toes. Pathologic or high trauma fractures were also excluded.
ARR = absolute risk reduction.