EVENITY® was compared to a commonly prescribed antiresorptive2
Phase 3 Event-Driven Study in Postmenopausal Women With Osteoporosis Receiving EVENITY® First Followed by Alendronate vs Alendronate Alone1,2
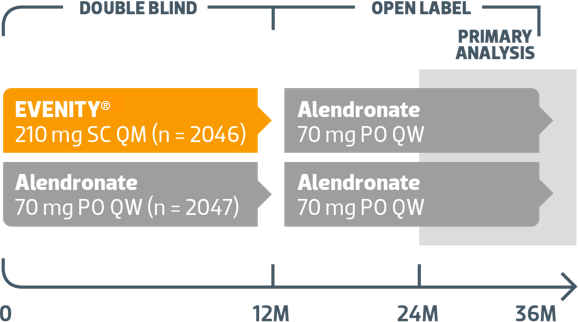
SC = subcutaneous; PO = orally; QM = monthly; QW = weekly.
Randomized, double-blind, alendronate-controlled, event-driven study1
- Postmenopausal women, 55-90 years old
- Bone mineral density (BMD) T-score ≤ -2.5 at total hip or femoral neck, and
- Either one moderate or severe vertebral fracture or two mild vertebral fractures
- Postmenopausal women, 55-90 years old
- BMD T-score ≤ -2.0 at total hip or femoral neck, and
- Either two moderate or severe vertebral fractures or a history of a proximal femur fracture
OR
4093 women randomized to receive 12 months of treatment1
- Monthly subcutaneous injections of EVENITY® (n = 2046), or
- Weekly oral alendronate (n = 2047)
All women supplemented with daily calcium and vitamin D
After 12-month treatment period, women in both arms transitioned to open-label alendronate while remaining blinded to their initial treatment
Co-primary endpoints1
- Incidence of morphometric vertebral fracture at 24 months
- Time to first clinical fracture (nonvertebral and symptomatic vertebral fracture) through the primary analysis which was performed when at least 330 subjects had a clinical fracture and all subjects had completed the 24-month visit
