EVENITY® compared to the most commonly used bone builder through 12 months1
Phase 3 Study in Postmenopausal Women With Osteoporosis Receiving EVENITY® vs Teriparatide1
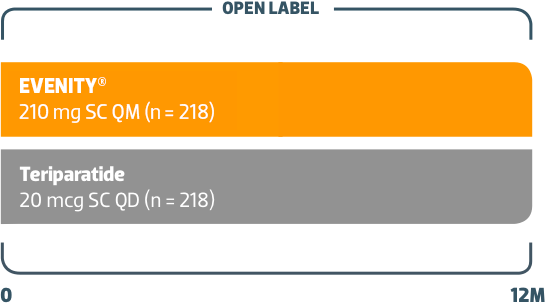
SC = subcutaneous; QM = monthly; QD = once daily.
Randomized, open-label study1
- Postmenopausal women, 55 to 90 years old
- Had osteoporosis and history of nonvertebral fracture after age 50 or vertebral fracture at any time
- Patients were receiving oral bisphosphonates at least 3 years prior to enrollment
436 women transitioned from bisphosphonate therapy to1
- EVENITY®, or
- Teriparatide
All women supplemented with daily calcium and vitamin D
Primary endpoint1
- Percent change from baseline in total hip BMD through month 12 (mean of months 6 and 12)
Consider open-label study limitations when interpreting results. This open-label study was not blinded.
